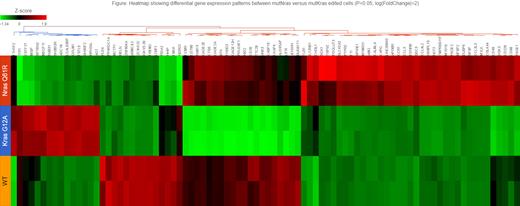
Myeloma is well known to have significant genetic heterogeneity among patients' tumors, affecting both progression as well as therapeutic responses. Moreover, subclonal diversity can lead to selective outgrowths that are influenced by new mutations, as well as the bone marrow microenvironment and therapeutic exposures. The ras family of genes (N -and K-) are the most common mutations (40-50% ) in myeloma. Notably, ras mutations occur among a variety of other co-existing chromosomal abnormalities. Ras mutations were found to be more commonly associated with cyclin D1 high expression (e.g., resulting from 11;14 translocations). We and others have also shown that rasmutations may not be disease-initiating but may arise during disease progression and relapse; or are subclonal events that impart selective growth advantage and emerging drug resistance. There is evidence that activating mutations (codons 12,13, 61) in K- ras may have different clinical impact than activating mutations in N- ras yet there are conflicting clinical outcomes that may be due to therapies analyzed or differences in other co-existing genetic mutations. Previous studies using transfection of genes with targeted mutations show random integration and often in multiple copies, thus, limiting interpretations. We chose to create N ras61 and K ras12 mutations by CRISPR-cas directed edits in a single endogenous ras allele in a myeloma cell line (FLAM76) that initially had two wild type alleles (and an 11;14 translocation). We then compared the impact of these edits on growth, signaling, gene expression, and drug responses. We find both the mutN ras and mutK ras edited cells have increased proliferation compared to wild type. However, we find that the N ras mutation increases MAPK signaling and downstream gene activation to a greater extent than the K rasmutation. Based on RNAseq analysis, a MAPK gene activation score was higher in mutN ras edited cells than mutK ras edited cells. Further, we see distinct gene expression patterns between mutN ras versus mutK ras edited cells (See heat map Fig) impacting distinct pathways. Notably, mutNras cells have much higher NOXA expression, increasing sensitivity to apoptosis-inducing drugs (e.g., proteasome inhibitors); and Kras induces anti-apoptotic Bcl-xL, reducing sensitivity. We have conducted a high throughput drug screen with 84 drugs, and while some drugs are active against both mutN ras and mutK ras edits (Proteasome inhibitors), we have identified drugs with differential response to K versus N ras mutations. For example, mutK ras FLAM76 cells are more resistant to proteasome inhibitors; and particularly more resistant to HSP inhibitors and HDAC inhibitors compared to wild type or mutN ras cells. In contrast, mutK ras cells are more sensitive to PikFyve and IL-12 inhibitors than mutNras or wild type cells. Gene expression analysis shows that mutKras cells result in more downregulation of apoptotic genes and higher upregulation of cell survival genes, as well as ER stress pathway genes. Our results suggest that treating myelomas containing K versus N ras activating mutations may benefit from understanding distinct pathway activations and targeting selective drug therapies. We are currently testing new pan-Ras inhibitors to determine if there are distinct responses between our N- and K- ras edited cells, and we are expanding testing to a wider panel of myeloma cell lines containing ras mutations in the context of other genetic abnormalities. Details of gene expression distinctions, pathways, and drug responses that distinguish mutN ras and mutK ras in myeloma will be presented.
Disclosures
No relevant conflicts of interest to declare.